β-lactamases are enzymes produced by bacteria (also known as penicillinase) that provide multi-resistance to β-lactam antibiotics such as penicillins, cephamycins, and carbapenems. TEM-1 is the most commonly encountered beta-lactamase in Gram-negative bacteria. Although TEM-type beta-lactamases are most often found in E. Coli and K. Pneumoniae, they are also found in other species of Gram-negative bacteria with increasing frequency. The amino acid substitutions responsible for the extended-spectrum beta-lactamase (ESBL) phenotype cluster around the active site of the enzyme and change its configuration, allowing access to oxyimino-beta-lactam substrates. Opening the active site to beta-lactam substrates also typically enhances the susceptibility of the enzyme to β-lactamase inhibitors, such as clavulanic acid. Single amino acid substitutions at positions 104, 164, 238, and 240 produce the ESBL phenotype, but ESBLs with the broadest spectrum usually have more than a single amino acid substitution. Based upon different combinations of mutations, currently, 140 TEM-type enzymes have been described.
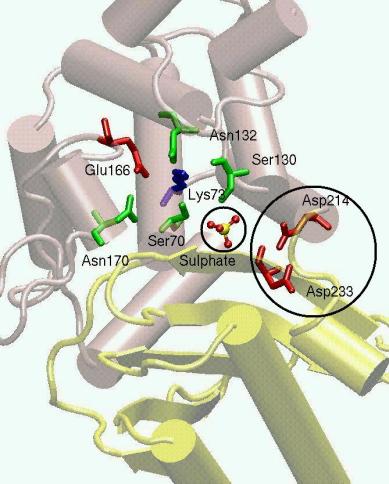
Figure 1: Crystal structure of the TEM-1 betalactamase in secondary structure representation. The residues in the active site are evidenced. The aspartic residues in the circle form a hydrogen bond stabilizing interaction.
We have investigated the dynamical properties of the TEM-1 β-lactamase by MD simulations [1]. In particular, the effect of the protonation of the aspartyl dyad D214-D233 on the structural and dynamical properties of TEM-1 -lactamase in aqueous solution was investigated. MD simulations of free enzyme were performed assuming the aspartyl dyad either in the mono- and deprotonated state. The results show that when the dyad is mono-protonated, the two domains remain tightly packed, as in the crystal. At variance, the deprotonated dyad destabilizes the two hinge regions generating the active site, and has effects on the conformation of the catalytic site residues (S130 and K234).
Figure 2: Essential modes visualizer: concept interface.
REFERENCE
- Roccatano, G. Sbardella, M. Aschi, G. Amicosante, C. Bossa, A. Di Nola, F. Mazza. Dynamical aspect of TEM-1 β-lactamase. J. Comp. Aided Mol. Des., 19, 329-340 (2005).
