The Hückel molecular orbital method is a quantum mechanics approach for calculating the energies of molecular orbitals of π electrons in conjugated hydrocarbon systems, such as ethylene, benzene, and butadiene. In this series of articles, I have summarized the main aspects of the theory with practical examples of applications and programming of the method.
The method was devised by Erich Hückel in 1930 and subsequently expanded and improved by other scholars. The technique has helped obtain important theoretical results in studying of the properties of organic molecules and the mechanisms of their chemical reactions in the period preceding the development of electronic computers. For example, he provided the theoretical basis of Hückel’s rule for the aromaticity of (4n + 2) π cyclic planar systems of electrons.
Hückel assumed that the electrons in bonds can be treated separately from those involved in
bonds for unsaturated organic molecules. The symmetry of the two types of molecular orbitals in which the electrons for the two types of bonds are located justifies this assumption. In fact, a
orbital is antisymmetric by reflection on the molecule plane, while a $ latex \sigma $ is symmetrical.
Hückel assumed that the π electrons can be treated separately by those involved in the bonds for unsaturated organic compounds. In fact, a π orbital is antisymmetric for reflection through the plane of the molecule, whilst a
one is symmetric.
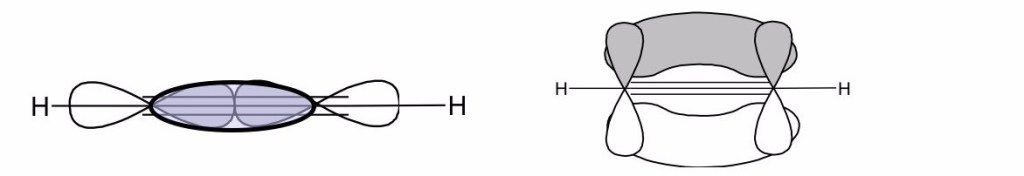
With such an assumption, we can then write the total energy of the molecular system as . This means that the wavefunction of the molecule is given by the product of the wavefunction describing the
and
electrons:
We can also assume that the molecular wave function of the whole system can be approximated as a product of 1-electron wave function orbitals
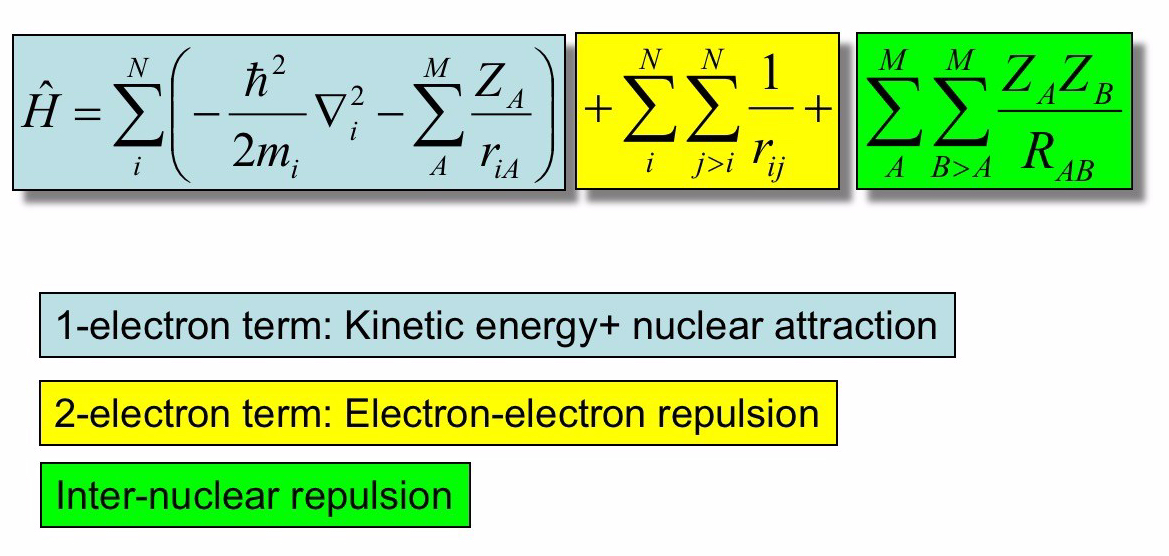
The energy of a sysmt can be evaulated using tha Hamiltonian quantum operator that is defined as
with
: the kinetic energy of the nucleus.
: the kinetic energy of the electrons.
: the proton-electron attraction potential energy.
: the proton-proton repulsion potential energy.
: the electron-electron repulsion energy.
In the case of a molecule composed by M atoms, the total hamiltonian can be written as
For a molecular sytem with π electrons, we can further distinguish these electron from the ones as

For not interaction electrons, we can also assume that the total energy of the system can be calculate by a total Hamiltonian (see my blog on the classical mechanics) operator can be expressed as
and the eigenstates of each electron can be calculate by the Schrödingen’s equations
As further assumption the wavefunction of the electrons can be factorized as follow
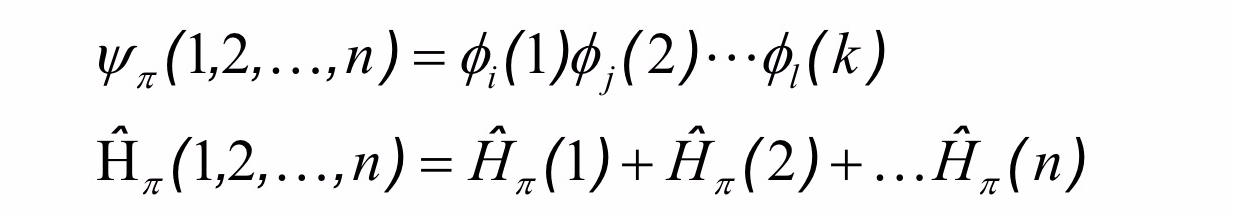
Using this approximations the total electronic energy from the electrons can be expressed as
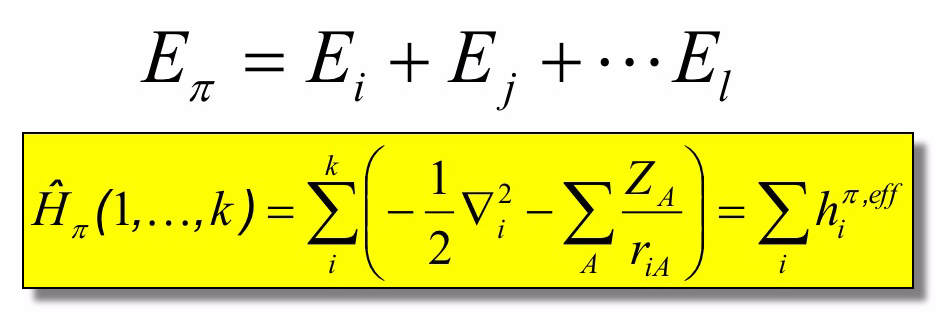
We are now going to consider a simple molecular system (the allyl molecule) to show how to assign the atomic orbitals to construct the Hückel determinant. An allyl molecule has the structural formula and consists of a methyl group bridge (
) attached to a vinyl group (
). The name of the molecule derives from the Latin word for garlic, Allium sativum, and was chosen by Theodor Wertheim in 1844 when he succeeded in isolating a derivative from garlic oil.
We consider only the three orbitals of each carbon since they are antisymmetric for reflection along the molecular plane. The total molecular orbital can be considered as a linear combination of the atomic orbitals.
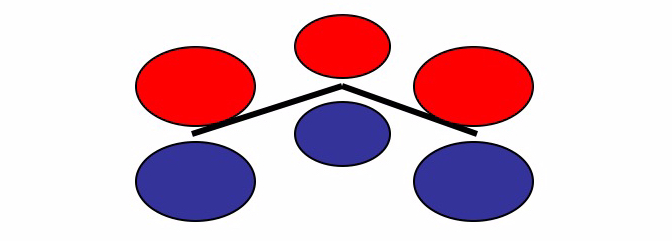
The total molecular orbital can be considered as a linear combination of the atomic orbitals.
In order to find the energy of the molecular system, we are going to use the result of the so called variational theorem.
The variation theorem states that given a system with a Hamiltonian operator then if
is any well-behaved function that satisfies the boundary conditions of the Hamiltonian, then

where is the true value of the lowest energy eigenvalue of
This expression is also called Rayleigh ratio.
The variational principle allows us to calculate an upper bound for the ground state energy by finding the trial wavefunction for which the integral is minimised. The principle assure us that only if the function is exact
in all the other cases
.
Linear variation method
We are going to use the so-called linear variation method (or Rayleigh-Ritz method) in which the linear variation function is a linear combination of linearly independent atomic orbital functions that satisfy the boundary conditions of the problem (
). The coefficients (
) of the trial wavefunction are parameters to be determined by minimising the variational integral.
By inserting the function of the allyl molecule in the Rayleigh ratio, the numerator become
where is the element
of the Hamiltonian matrix, and the denominator
where is the element
of the overlap matrix.
Therefore we can now write
or in the rearranged form as
(1)
Now, we need to find the value of the coefficients that minimize the function
using the variational method. This can be obtained using the definition of a mimimum of a function, namely by calculating the equations
for all the functions i.
By differentiating both the sides of (1) and using the properties of and
and of the coefficients
, we finally obtain the so-called secular equation
for all the k.
We therefore have a set of k simultaneous homogeneous equation (secular equations) in k unknowns. In the case of the allyl system the system is the follwing
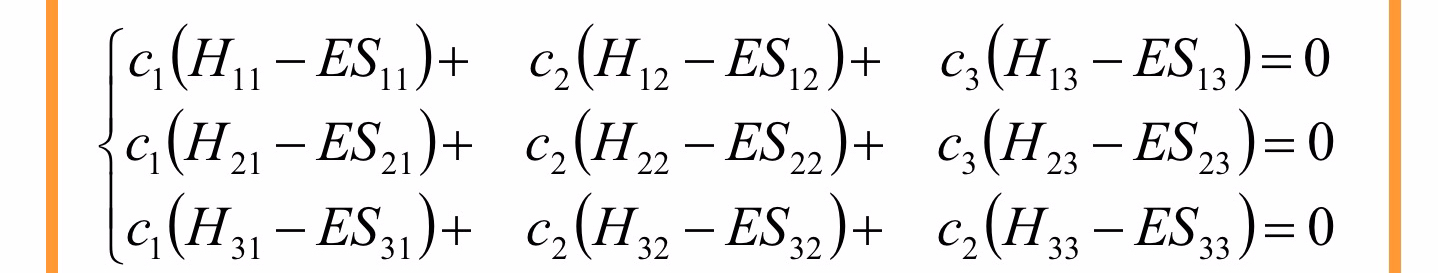
For a non-trivial solution (i.e. for all i), the determinant of the secular matrix must be equal to zero


Therefore we can set up the values of integral in Hückel determinant as constants and on the bases of the molecular connectivity as

and for the overlap integral
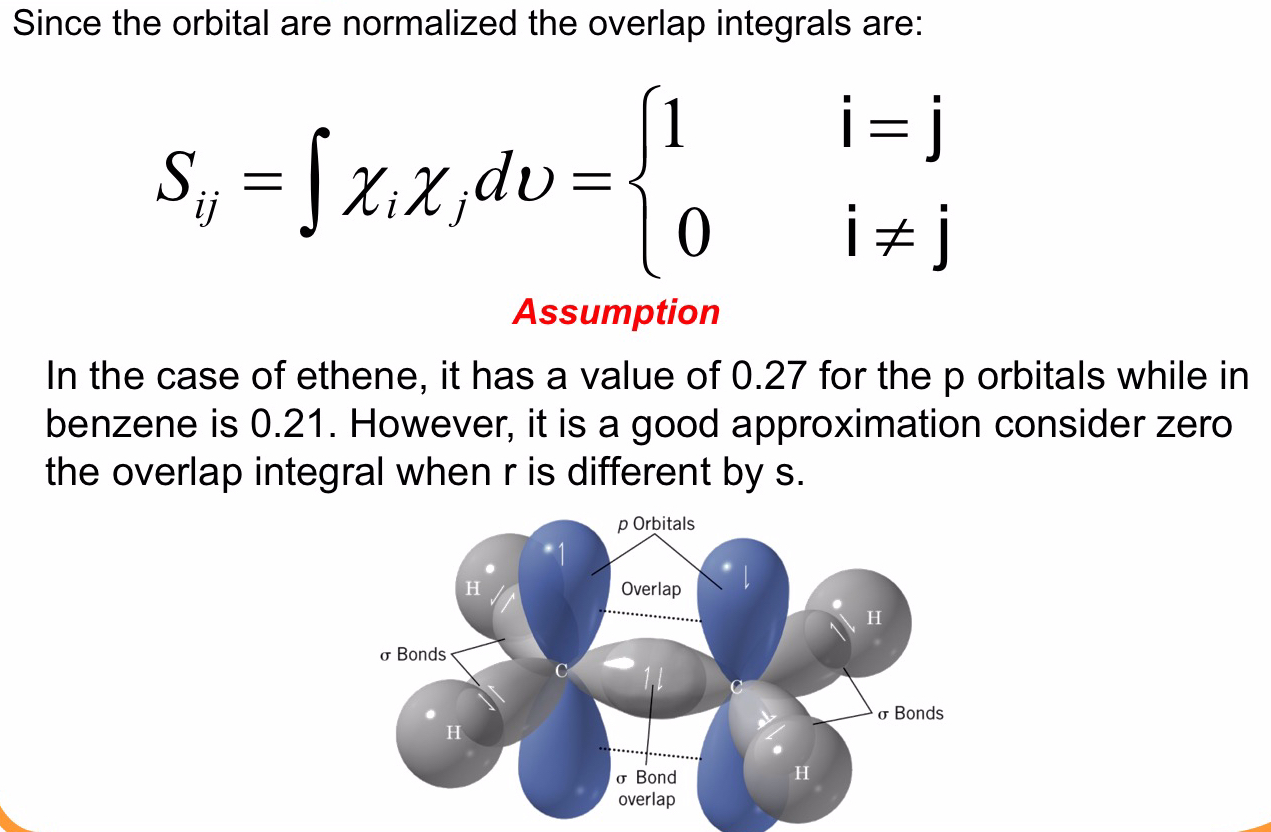
With these semplification the Hückel determinant for the allyl molecule become


Here some more example of Hückel determinat for simple unsaturated molecules
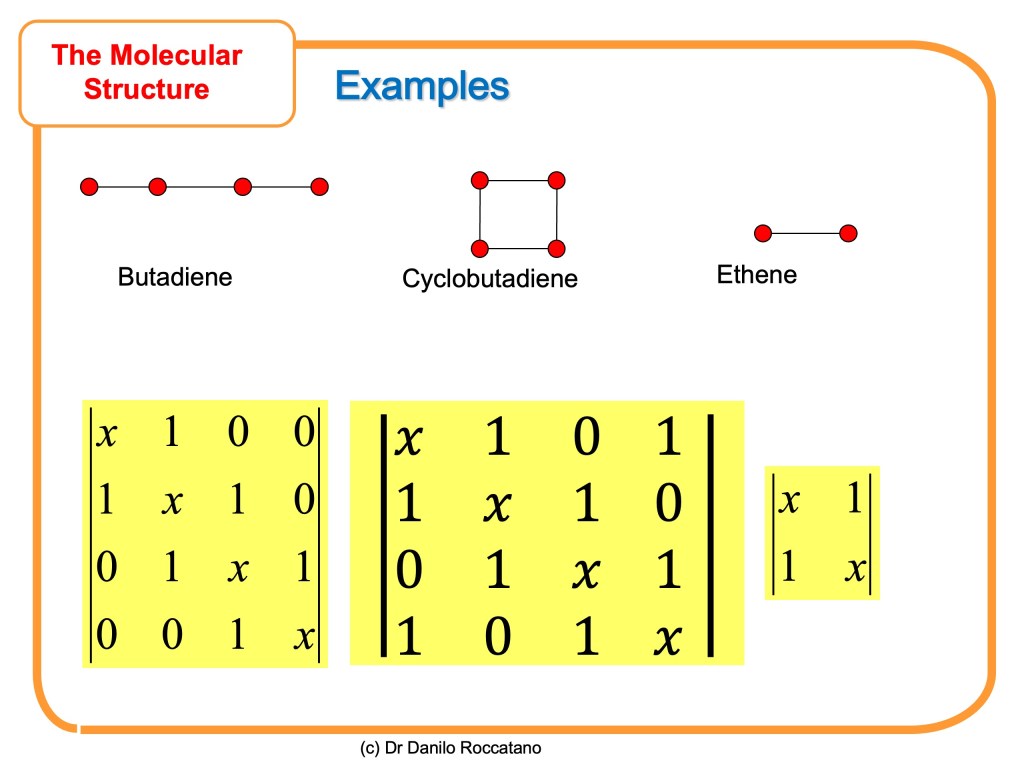
In the second part of this article, we are going to learn how to solve this determinantal equation to find the energy of the molecular orbitals.
If you have found useful this article, please tell your friends and do not forget to Like it and subscribe for updating!
REFERENCES
J.P. Lowe Quantum Chemistry. 1993, Academic Press.

Pingback: Physical Chemistry: The Simple Hückel Method (Part V) |
Pingback: Physical Chemistry: The Simple Hückel Method (Part VI): PREVIEW |
Hello,
first thing thank you for the interesting articles on your web site.
Reading the Hückel Method (part I) I’ve spotted some mistakes.
In the sentence: ” In fact, a \pi orbital is antisymmetric by reflection on the molecule plane, while a $ latex sigma $ is symmetrical.” reported at the end of the third paragraph, the HTML page doesn’t show the sigma character but its latex code instead.
in slide11-e1605347034130.jpeg the error is that:
the second term in the first row of Hückel determinant for a three p(z) orbitals system is the same as the first term of the first line. It should be H12 – ES 12.
and
in slide17.jpeg there is an error in the Hückel determinat for cyclobutadiene
The correct determinant should be:
| x 1 0 1 |
| 1 x 1 0 |
| 0 1 x 1 |
| 1 0 1 x |
I hope this could help improve the quality of your web page.
Cheers,
alessandro
LikeLike
Hi Alessandro,
Thank you very much for reporting the mistakes. I am also happy to read that you find my articles interesting, and constructive comments are always welcome.
Cordiali saluti
Danilo
LikeLike