In the previous parts, we learned how to set up the Hückel determinant for conjugated linear and cyclic molecules and calculate the energy and coefficients of molecular orbitals. In this new article, we will use the SHM to calculate different molecular properties. These properties help understand the structure and reactivity of organic molecules.
CALCULATION OF THE pi-ELECTRON DENSITY
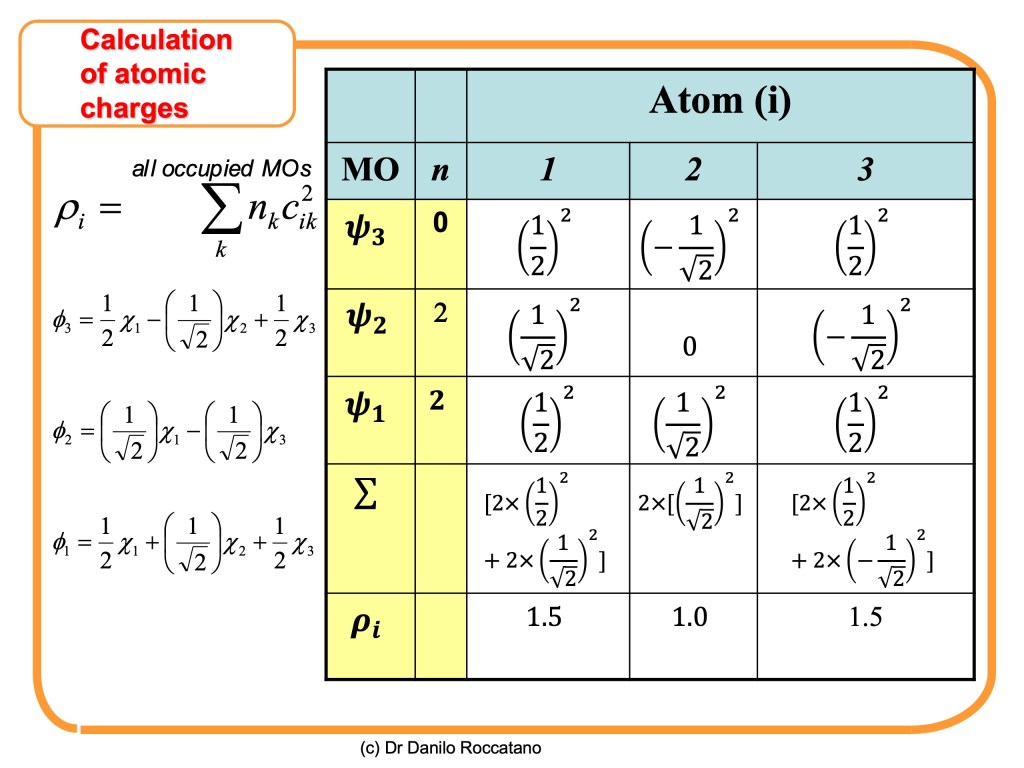
The electron density quantifies the amount of electronic charge localized around each atom in the molecule. This information gives a valuable estimation of the electrostatic (partial) charge on each atom of the aromatic system. The concept of fractional charge seems to undermine the idea of an electron as a particle. Still, it is a consequence of the probabilistic interpretation of the quantum realm. The Copenaghen interpretation of quantum mechanics theory states that the square of the wave function, associated with the description of the quantum particle’s wave nature, provides a probability density function (PDF). Whose multiplication with the infinitesimal volume of space produces a probability of localizing the particle in that volume. Therefore, the squared orbitals functions obtained by the SHM can be used to get the amount of negative charge on each atom of the aromatic system. For this purpose, we can sum the square of wave function coefficients of all the occupied orbitals
(1)
where is the number of electrons in the
orbital.
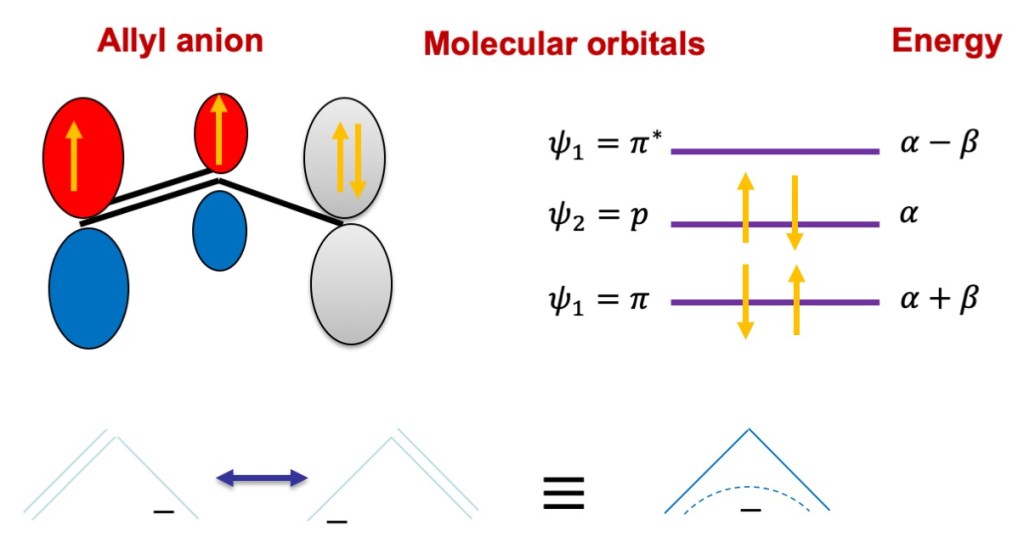
The Figure above shows the molecular orbitals for the allyl anion. Let’s now calculate the electron densities on the atoms of these molecules using the formula given before.
CALCULATION OF THE BOND ORDER
The bond order is another valuable property that can easily be derived from the HMO coefficients. It gives tha $#\pi-$$electron distribution in the bonds instead of on the atoms. It is defined as
In the following Figure, the calculation of of the allyl radical is reported. The result shows an equal contribution of 0.707 for both bonds.

The total bond order is obtained by adding the sigma bond (1) to as shown in the examples reported in the figure below.

For hydrocarbons, there is a correlation between bond order and bond lenght that can be used to make an approximate prediction of bond lenght variations. C.A.Coulson in article of 1939 has proposed the following formula that can be used to estimate the effect of the bond order on the length variation of a single bond
$$R=s-\frac{s-d}{1+\frac{k(1-p)}{p}}$$
where s is the average single bond length taken equal to 1.54 A for C-C, d, the double bond length equla to 1.337 A, p the bond order, and k an adjustable parameter. For the example given before, gor p=0.707 and k=0.795, the value of R is
$$R=1.54-\frac{1.54-1.337-d}{1+\frac{0.795(1-0.707)}{0.707}}=1.54-0.153=1.387 \AA$$
CHEMICAL REACTIVITY INDECES
CALCULATION OF THE FREE VALENCE
Finally, the free valence is defined as the difference between the maximum possible bond order and the actual tital bond order. It is defined as

UNDER CONSTRUCTION
If you have found interesting and useful my article, do not forget to press “Like it” and subscribe for updates on new ones!
REFERENCES
- J. P. Lowe. Quantum Chemistry. 1993, Academic Press.
- F.A. Carroll. Structure and Mechanism,1998, BROOKS/COLE Publishing Company.

Pingback: Physical Chemistry: The Simple Hückel Method (Part V) |
Pingback: Physical Chemistry: The Simple Hückel Method (Part VI): PREVIEW |