Myoglobin is a small ancient protein of ~150 amino acids specialized in the transport of respiratory gases to muscular tissues and back to the lungs of the superior organisms. Even though, it was one of the first protein having the three-dimensional structure solved using X-ray crystallography, the detail of the transport mechanism of gases in and out the active site is yet not completely understood. More recently, time-resolved x-ray studies have provided clues on the possible pockets in the protein interior where gases can transit but the detail of the migrations dynamics is unclear.
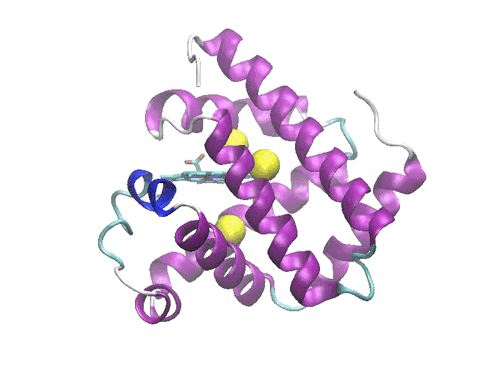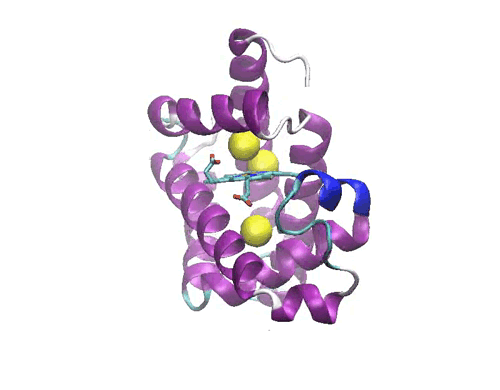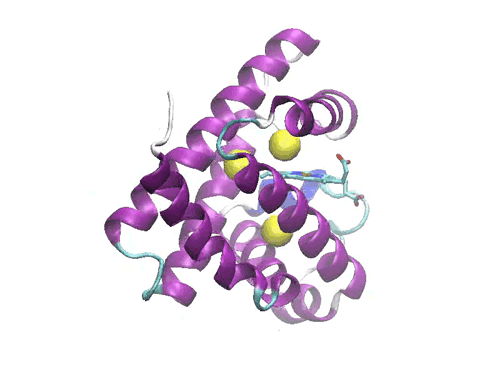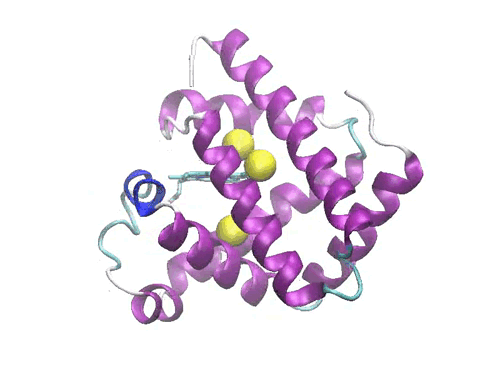
Figure 1: Crystal structure of the myoglobin (PDB code: 2w6x)bound to Xe atoms )in yellow). The Xe1 binding site is the one above the heme.
In a molecular dynamics study, we have investigated the diffusion of the photodissociated carbon monoxide in Sperm Whale myoglobin. The results evidence one possible ligand migration dynamics from the distal pocket to the Xe1 cavity (see Figure 1) via a path involving the other xenon binding cavities, and momentarily two additional packing defects along the pathway. Although our data refer to a single trajectory, the local dynamics of the ligand in each cavity is sufficiently equilibrated to obtain local structural and thermodynamic information. Free energy calculations of the migration between adjacent cavities show that the migration is not a simple diffusion, but is kinetically or thermodynamically driven by the collective motions of the protein. Comparison with recent time-resolved structural data obtained by Laue crystallography from few hundred picoseconds to milliseconds resolution range shows a good agreement with the X-ray determined snapshots. The simulation allows proposing some interesting hypothesis on the structural dynamics of Mb which are not accessible to crystallography. The compatibility between time-resolved crystallographic experiments and MD simulations paves the way to a deeper understanding of the role of internal dynamics and packing defects in the control of ligand binding in Haem proteins.
- C. Bossa, M. Anselmi, D. Roccatano, A. Amadei, B. Vallone, M. Brunori, A. Di Nola. Extended molecular dynamics simulation of the carbon monoxide migration in sperm whale myoglobin. Bioph. J., 86, 3855-3862 (2004).
