I have recently contributed to a proof of concept study published in the prestigious Nature Communication (doi:10.1038/s41467-018-03931-4) [1]. The study involved collaborations with experimental groups across the University of Lincoln (UoL), University of Molise (Italy) and the Royal Holloway University of London coordinated by Dr Enrico Ferrari (UoL).
The motivation for this is that, generally, the high diversity of protein properties necessitates the development of unique nanoparticle bioconjugation methods, optimized for each different protein. Enrico had the smart idea to design a universal bio-conjugation approach which makes use of a new recombinant fusion protein combining two distinct domains. For this purpose, the N-terminal part is Glutathione S-Transferase (GST, a protein ubiquitously present in both eukaryotes and prokaryotes with the ability to catalyze the conjugation of the reduced form of glutathione (GSH) to xenobiotic substrates for detoxification) from Schistosoma japonicum was bound covalently bound to gold nanoparticles (GNPs) by gold-sulfur bonds. The C-terminal part of this multi-domain construct is the SpyCatcher from Streptococcus pyogenes, which provides the ability to capture recombinant proteins encoding a SpyTag (see Figure 1).

The result of this work has shown that Spy- Catcher could be immobilized covalently on GNPs through GST without the loss of its full functionality. We then show that GST-SpyCatcher activated particles can covalently bind a SpyTag modified protein by simple mixing, through the spontaneous formation of an unusual isopeptide bond.
In this paper, we have used Molecular Dynamics (MD) simulations to study the absorption of the protein on the surface of the gold nanoparticles coated with citrate molecules. In the simulation, a flat gold surface is used to approximate the nanoparticle surface. Atomistic simulation of the entire nanoparticle would be challenging since the NP used in the experimental study (~40 nm in diameter) is too large (only the NP would consist of 2 millions of atoms, see Figure 2).

The Au nanoparticles form by reduction of gold salts in a citrate solution whose concentration determines the diameter of the nanoparticles. The citrate molecules are absorbed on the nanoparticle surface and negatively charge it. In the MD simulations, we have also used a model of Au surface coated with physioabsorbed citrate molecules. In this way, the citrate molecules in complex with Na counterions coat the gold surface as a “molecular” quill (see Figure 3)!

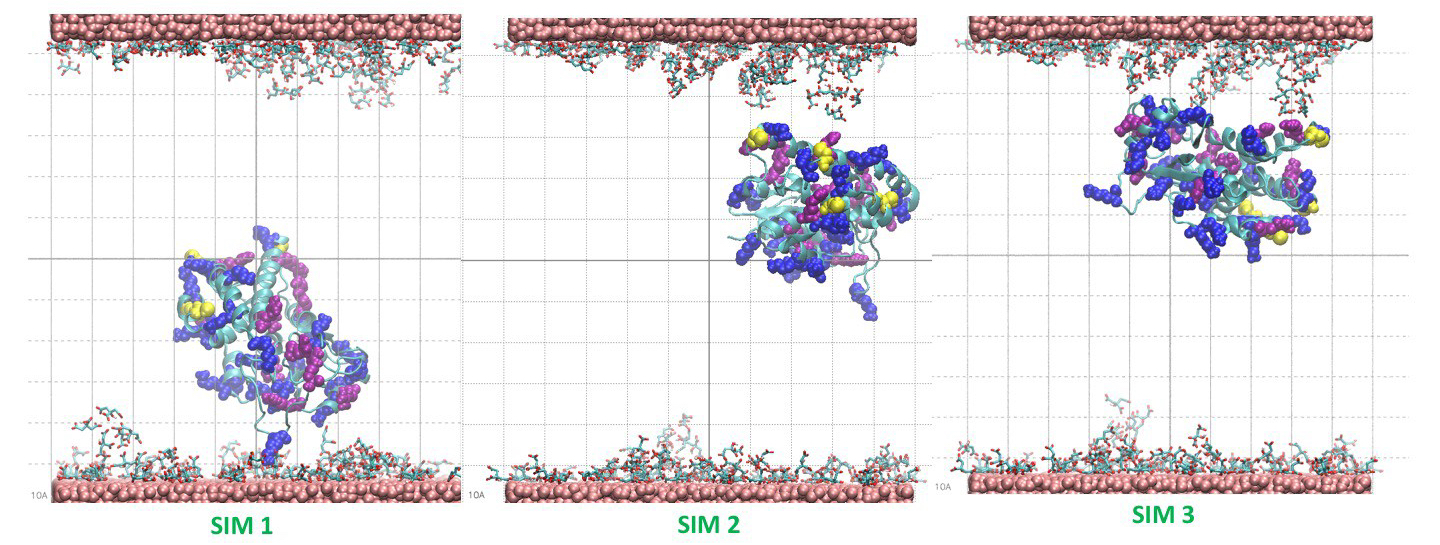
The result of the simulations indicates the tendency of the protein to bind the coated layer driven by electrostatic interactions with the negative carboxylates of the citrate with the side chain of the arginine residues on the protein surface. Figure 4 shows the conformations of the protein bonded to the layer at the end of three 20 ns simulations starting from different velocities. The results support the experimental finding on the possible mechanism of absorption of the protein on the nanoparticles.
REFERENCE
- Ma, A. Saccardo, D. Roccatano, D. Aboagye-Mensah, M. Alkaseem, M. Jewkes, F. Di Nezza, M. Baron, M. Soloviev, E. Ferrari. Modular assembly of proteins on nanoparticles. Nature Communication.9, 1489 (2018).
