The recent progress on time-resolved optical spectroscopy and FRET techniques have opened new exciting perspectives to the understanding the short timescale dynamics of peptides in solution. In fact, experimental data provided by these measurements can be directly compared with the results of computer simulations.
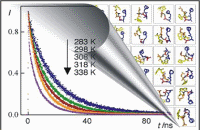
In particular, the end-to-end contact kinetics of short peptide in solution has been extensively investigated. These studies provide important information on the kinetics of loop formation that several theoretical and experimental evidence has shown to be the fundamental step in driving the peptides or proteins folding. Therefore, the accurate determination of the time scale on which these events occur can shade a light on the protein folding kinetics. Furthermore, the time scale of those processes is in the order of few tens of nanoseconds, that make possible, for the first time, a direct comparison with atomistic simulations. The kinetic behavior of different peptides having the Tryptophan as the quencher and an amine derivative of 2,3-diazabicyclo[2,2,2]oct-2-ene (DBO) linked to the end Aspartic amino acid, as the fluorophore has been investigated. It occurs that this fluorophore has an unusually long life singlet excited state in aqueous solution ( up to 0.5 microseconds), and high solubility in water. Thus, the tryptophan/DBO forms a unique probe/quencher pair characterized by a contact quenching mechanism, which differentiates from conventional fluorescence resonance energy transfer donor/acceptor pairs. This offers the possibility to compare direct time-resolved kinetic data with the indirect kinetic information extracted from steady-state fluorescence intensity measurements.
REFERENCES
- Roccatano, W. M. Nau, M. Zacharias. Structural and dynamic properties of CAGQW peptide in water: a molecular dynamics simulation study using different force fields. J. Phys. Chem. B, 118, 18734-18742 (2004).
- Sahoo, D. Roccatano, M. Zacharias, W. M. Nau. Distance distributions of short polypeptides recovered by fluorescence resonance energy transfer in the 10 Å Domain. JACS, 128, 8118-8119 (2006).
- Roccatano, H. Sahoo, M. Zacharias, W. M. Nau. Temperature dependence of looping rates in a short peptide J. Chem. Phys B, 111, 2639-2646 (2007).
- Sahoo, D. Roccatano, A. Henning and W. M. Nau. A 10 Å spectroscopic ruler applied to short polyprolines. JACS, 129, 9762-9772 (2007).
